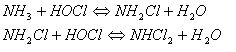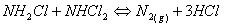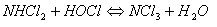|
Index page |
|
|
|
TASK 2 HEALTH EFFECTS OF CHLORINE Some researchers say that chlorine has some very serious health consequences when used as a sanitizer in swimming pools. Chlorine is a highly reactive gas. Effects of chlorine on human health depend on the concentration of chlorine present in air, and on the duration and frequency of exposure. They also depend on the health of an individual and the environmental conditions during the exposure. Chlorine enters the body breathed in with contaminated air or when consumed with contaminated food or water. When small amounts of chlorine are breathed in during short time periods, it can affect the respiratory system (coughing, chest pains, and fluid accumulation in the lungs). Chlorine can also cause skin and eye irritations. But these effects do not take place under natural conditions. Besides, chlorine is not very persistent in the body, due to its reactivity. However, pure chlorine is very toxic and can be deadly at even small amount (chlorine gas in World War I).
1) Chloramines:
But, during the last years, the issue of the chlorination of swimming pool has received considerable attention mainly with regard to the presence of chloramines. These substances are potentially carcinogenic or teratogenic chlorinated by-products. In fact, the smell and irritant properties of swimming pool air are mainly due to chloramines. Thus, when swimmers complain of too much chlorine in the pool area, the real problem is too little free chlorine in the water (too much chlorine has been converted to chloramines). The chloramine concentrations measured in the ambient air of the swimming pools would be 7 to 18 times higher than those of chlorine, which indicates that they are responsible of the typical odour of the halls of swimming pool. These chloramines, monochloramine (NH2Cl), dichloramine (NHCl2) and trichloramine (NCl3) are generated from the reaction of HOCl with ammonia and amino-compounds that originate from sweat and urine of the swimmers.
They are (especially the trichloramine) quite volatile and they partition easily from the water into the air. The main factors that determine the production and the air levels of these by-products are: the degree of water chlorination, the contamination of the water by nitrogen-containing compounds (which depends on the number of bathers, their behaviour and hygiene), water temperature and air recirculation. Chloramines are mildly toxic to bacteria, but tend to be irritating to eyes and skin, so they are unwelcome substances in a swimming pool. Recent researches completed in 2002, by doctors in Fortunately, chloramines react with each other, as:
And if too much HOCl is available in the pool, an undesirable competing reaction takes place:
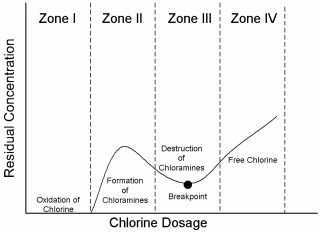
But nitrogen trichloride (NCl3) is very irritating to eyes and mucous membranes. Indeed, the study previously quoted, identified in laboratory challenge tests, that nitrogen trichloride levels of 0,5 mg/m3 were sufficient to trigger asthma type reactions in exposed persons. These levels are often exceeded in the air above indoor swimming pools. For these reasons, it is necessary to minimize chloramine formation in and above the swimming pools, by having swimmers wash before entering, by treating the water, by maintaining adequate water exchange rates, and by constant dilution of the air above indoor swimming pools via good ventilation practises. There are several water treatments to control chloramine formation. The best corrective action is to add more chlorine to the water. It is called “super-chlorination” or “shock-chlorination”. It consists in adding to the water for a short period of time, up to ten times the usual dose of chlorine. This treatment is illustrated by the diagram below:
Other chemical treatments are also available, for example the addition to the water of: · Sodium thiosulfate: it reacts with the chloramines to convert them back to ammonia and hypochlorous acid. · Potassium monopersulfate: it is a strong oxidizer with no chlorine content. It eliminates organic contamination, blocks chloramine production and enhances the efficiency of the existing chlorination.
2) Trihalomethanes (THMs) The trihalomethanes are also by-products of chlorination which can be harmless for the human health. They come from the substitution of the hydrogen atoms in methane (CH4) with the halogen atoms of chlorine or bromine. The methane is a natural product of our bodies and the products of this substitution called “THMs” are for example: ü CHCl3 Chloroform or trichloromethane ü CHBr3 Bromoform ü CHCl2Br Bromodichloromethane ü CHClBr2 Dibromochloromethane Like the chloramines, the rate of THM production is a function of the number of swimmers, the total organic carbon content of the water, the pH and the water temperature. The most important of the THMs is chloroform and chloroform as well as the dibromochloromethane, are suspected human carcinogens. Besides, swimming pools are considered to be the prime source of the public’s exposure to chloroform (over 70% after one hour of swimming). Chloroform can be measured in the blood or urine of regular swimmers. Exposure to low concentrations of chloroform causes renal and liver defects. The INRS (French National Institute for the Scientific Research) suggests not exceeding a concentration of 0,5 mg/m3 in the atmosphere of the halls of swimming pool. Epidemiological research shows there might be a relation between skin exposure to chlorinated organic substances and hypochlorite, and skin cancer. So it is essential to maximize the use of ventilation to remove these potentially harmful contaminants from the air of indoor pools. 3) Research on health effects of disinfection by-products ü Health research with swimming pool attendants: For a Dutch research, carried out in 2001, swimming pool attendants were interviewed. The research shows that a lot of employees suffer from forgetfulness, fatigue, chronic colds, voice problems, eye irritations, headache, sore throat, eczema and frontal sinus inflammation. Fertility problems were also mentioned. All problems are probably caused by working conditions: work during long hours in a warm and humid environment, and with exposure to chemical substances. When swimming pool attendants do not work, health problems vanish. Besides, in a French research undertaken by INRS, it has been shown that the first workers’ complaints appeared for a chloramine concentration of 0,5 mg/m3. All agents were complaining for a concentration of 0,7 mg/m3. The threshold values concerning the tastes and the odours of chloramines are respectively of: § Monochloramine: 0,65 mg/L et 0,48 mg/L § Dichloramine : 0,8 mg/L et 0,5 mg/L § Trichloramine : no available studies ü Health research with competitive swimmers Epidemiological researches have shown that competitive swimmers have more bronchial hyperresponsiveness or asthma than other sportsmen. Indeed competitive swimmers weekly spend many hours in the swimming pools. During their exercise, their physical effort is large and their inhalation is deeply and powerful. As a consequence, they inhale more air and absorb more chlorine products than recreational swimmers. But the complaints disappear when they swim in outdoor swimming pools, because wind removes gasses from the air above the pool. Besides, hypochloric acid (produced by chlorine and water) with sunlight can cause the pH value to drop. And when the pH value drops below 3,6 swimmers can suffer from dental abrasion: tooth enamel dissolves and the teeth become brittle and sensitive. ü Health survey of schoolchildren This survey has taken place in Besides, it is important to take into account that a one year old child would absorb in one hour in a chlorinated swimming pool on average three times more chloroform than a swimming teacher in one week, which would be higher than the tolerable daily dose fixed by WHO (World Health Organization).
You can send comments : aub@niras.dk
Started by NIRAS supervisor Sergio Fox on 27th March 2006. © COPYRIGHT 2001 ALL RIGHTS RESERVED Aurelie.dk |